Bispecific Gamma Delta-T-Cell Engager Directed to EGFR
PF-08046052 is an investigational compound. Its safety and efficacy have not been established.
Overview + Rationale
- EGFR is a cell surface receptor expressed in many cancer types.1
- γ9δ2-T cells are a conserved T cell subset that can induce cell death in a wide range of malignancies.2
- Activated γ9δ2-T cells directly induce cell lysis as well as activate downstream innate and adaptive immune cells, enhancing tumor cell killing.3-5
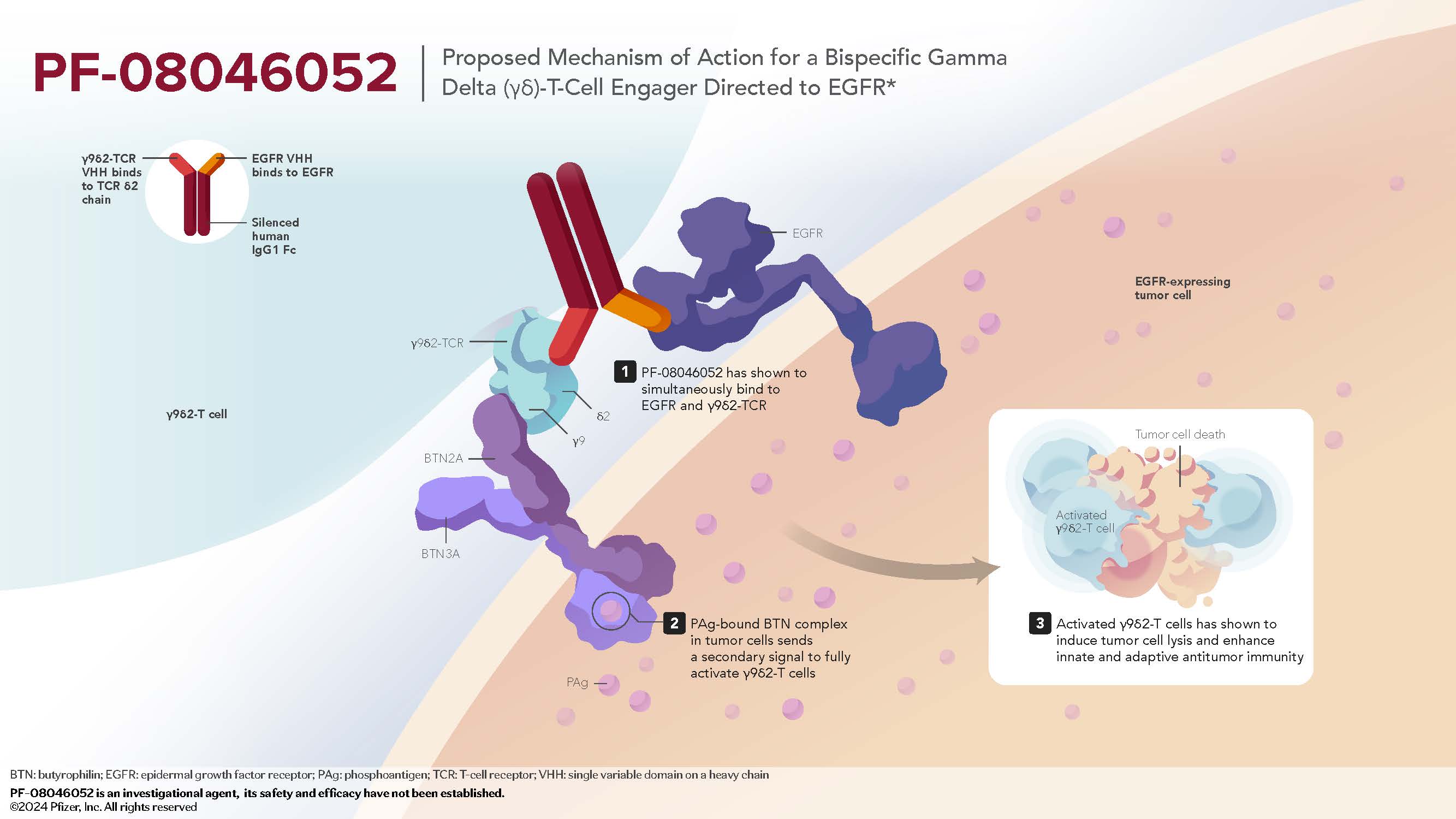
- PF-08046052 is a novel, investigational bispecific gamma delta (γδ)-T cell engager directed to EGFR, comprising a bispecific humanized heavy chain-only antibody designed to conditionally activate γ9δ2-T cells by simultaneously binding EGFR and γ9δ2 TCR.6
- Full activation of γ9δ2-T cells and tumor-directed cytotoxicity requires the presence of pAg, a stress signal found in malignant cells, but not in healthy cells.6,7
Mechanism of Action

 Back
Back
