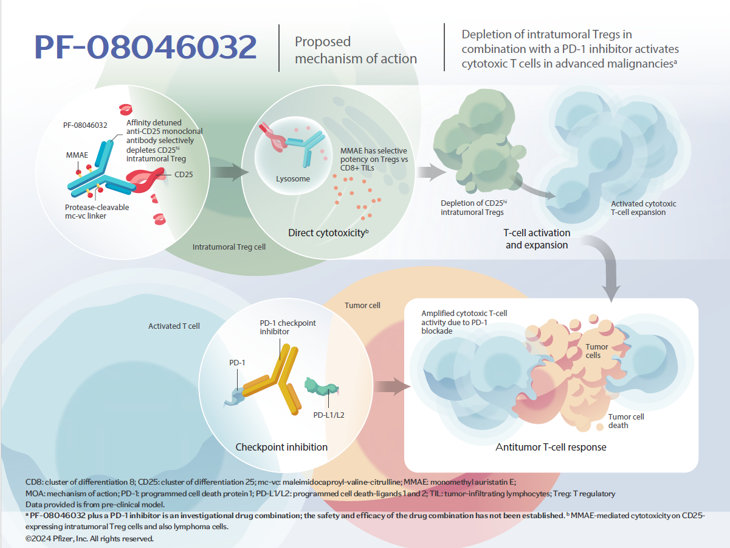
CD25V Directed Antibody Drug Conjugate
PF-08046032 is an investigational compound. Its safety and efficacy have not been established
Overview + Rationale
PF-08046032 is a vedotin antibody-drug conjugate (ADC) engineered to target and deplete immunosuppressive regulatory T cells (Tregs) within the tumor microenvironment (TME) through direct cytotoxicity.
PF-08046032 directs delivery of the cytotoxic MMAE (monomethyl auristatin E) payload to cells expressing CD25 via affinity-detuned binding of the alpha chain of the IL-2 receptor, which is highly expressed on intratumoral Tregs and lymphoma cells.
In vitro, affinity-detuning enables targeting of CD25hi intratumoral Tregs while sparing peripheral Tregs.
Mechanism of Action

 Back
Back

